Therapeutic IgY(ΔFc) Antibodies to MDR-Bacteria
Healthcare associated infections (HCAI) are persistent challenge for hospitals worldwide. A prevalence survey showed an average of 8.7% of hospital patients had HCAI. Patients who receive care in intensive care unit (ICU) are at increased risk for nosocomial infections; the emergence of antimicrobial-resistant pathogens in ICUs has made treating these infections very difficult and in some cases, impossible. Gram-negative bacilli are frequently associated with nosocomial infections in ICU patients. Of particular concern is the nosocomial infection caused by enterobacteria-propducing Extended-Spectrum Beta-Lactamase (ESBLs). Organisms that possess these enzymes are usually resistant to all currently available antimicrobials.
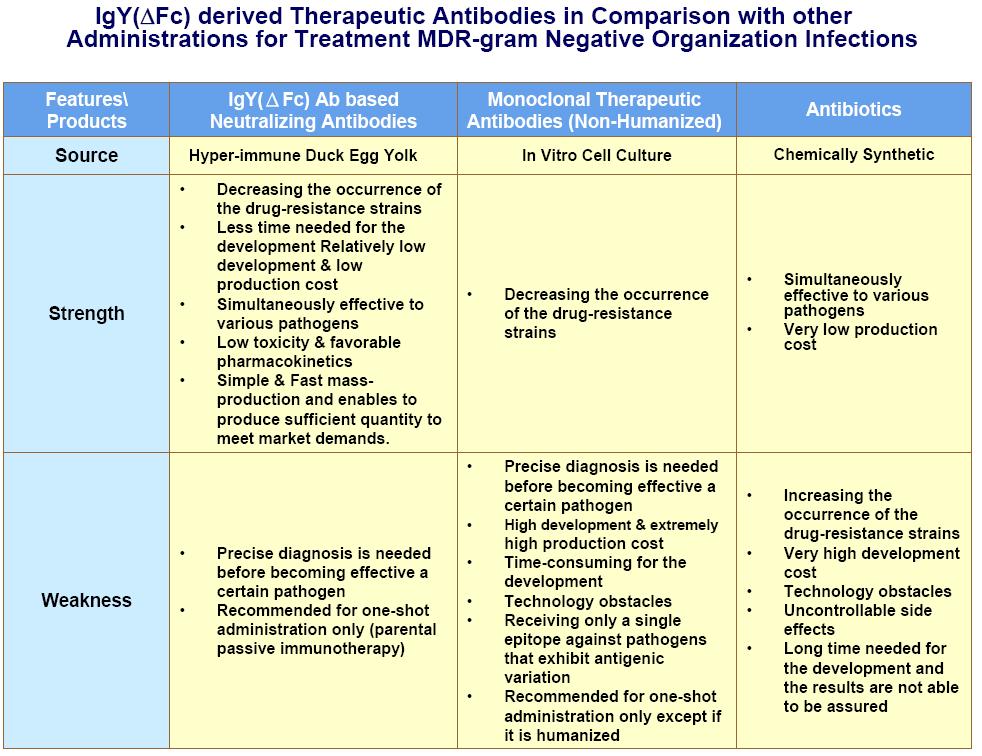
To address these threats, a number of different therapeutic strategies can be developed. The development of neutralizing antibodies against gram-negative bacilli (e.g. Pseudomonas aeruginosa, E coli) has the high potential to meet significant unmet medical needs. Passive administration with specific polyclonal antibodies or fragments target against disease causing agents has the advantages including:
- Offering clinical protection immediately after infection with pathogenic bacteria
- Versatility
- Low toxicity
- Favorable pharmacokinetic
- Time saving for the development
As for anti-bacteria therapeutic antibody, polyclonal antibodies are favored than monoclonal antibodies due to polyclonal antibodies providing polyvalent interactions that permit the application of therapeutic strategies against multiple epitopes or targets, which means polyclonal antibodies may target numerous biochemical pathways.
The unique advantages of IgY(ΔFc) antibody as well as the built cutting edge production technology are expected to be the key source of developing neutralizing antibodies for the treatment of nosocomial infections caused by multi-drug resistant gram-negative organisms (MDR-gram negative bacilli).
More information about partnering opportunities of IgY(ΔFc) Antibody derived Neutralizing Antibodies, please contact: partnership@good-biotech.com

